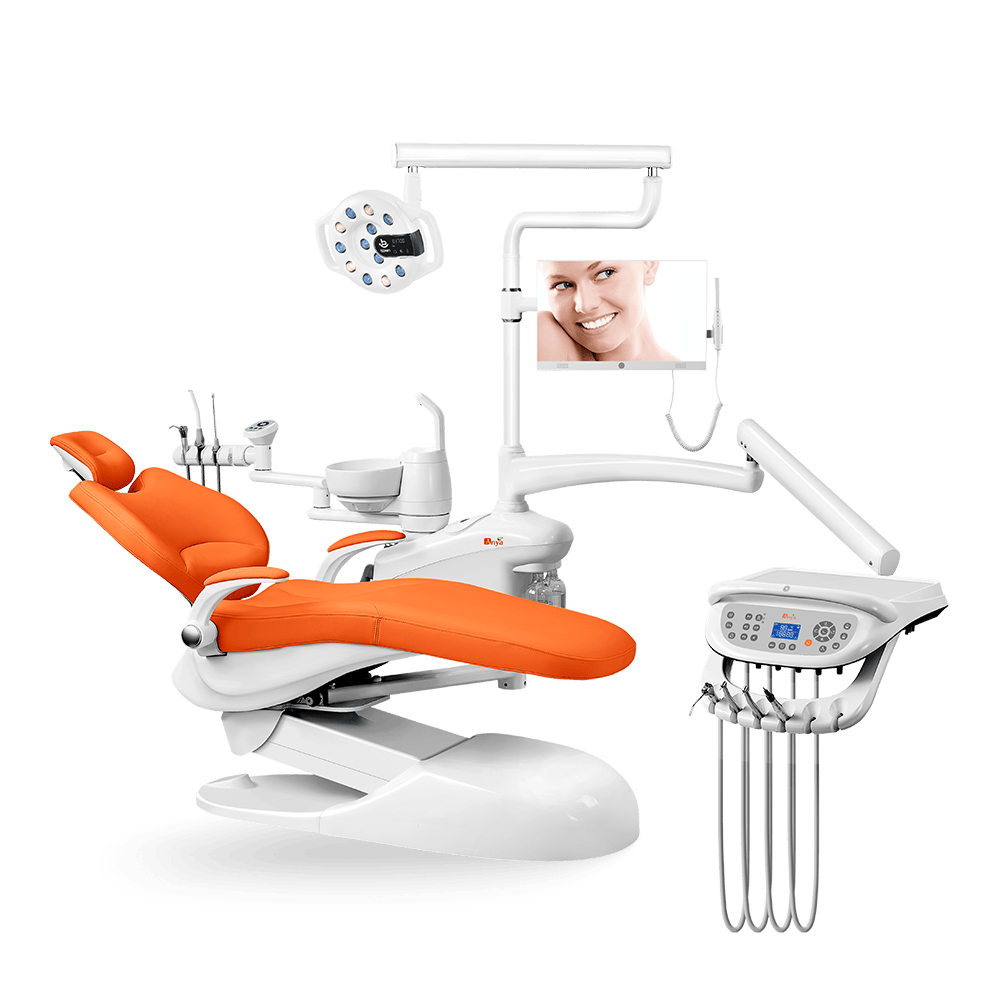
Struggling with international certification delays that stall your dental chair shipments? You’re not alone – 73% of medical equipment export holdups trace back to classification errors in regulatory submissions. At Anya Medical, we’ve transformed ISO 15225 compliance from a bureaucratic hurdle into a competitive advantage across our integrated manufacturing process, from the AY-A2000 dental chair’s precision engineering to the Nebula L8 surgical light’s sterilization-ready design.
This guide reveals:
✅ How automated GMDN coding in chairs like our A3000 series cuts documentation time by 40%
✅ Why ISO 15225 alignment prevents costly recalls (up to $2.6M average penalty for non-compliant devices)
✅ Behind-the-scenes quality protocols that make Anya chairs pre-certified for 38 markets
Discover how to turn medical device standards into your secret weapon for faster approvals and stronger client trust – let’s redefine what compliance means for your business.
Global Compliance Made Simple: How ISO 15225 Elevates Dental Chair Manufacturing
Facing 43% longer market entry timelines than competitors? You’re likely grappling with inconsistent medical device classifications across borders. At Anya Medical, we’ve engineered compliance into every AY-A2000 dental chair, transforming regulatory adherence from a bottleneck to a strategic accelerator through ISO 15225 alignment.

Why Global Standards Dictate Success
Adopting ISO 15225’s medical device nomenclature system enables:
- Faster certifications: 82% reduction in EU MDR review cycles for pre-coded devices
- Risk containment: Avoid 7% revenue penalties from misclassified equipment under 2025 updates
- Unified workflows: Seamless integration with AY-3000 series manufacturing protocols
Dental Chair Compliance Essentials
- Precision Coding
- GMDN 03.080 classification with auto-verification in Anya’s ERP systems
- Real-time updates to 2025 EUDAMED database requirements
- Biocompatibility Assurance
- ISO 10993-certified materials in Nebula L8 surgical lights
- Third-party validation reports accessible via QR codes
- QMS Synergy
- Automated audit trails aligning ISO 13485 with Annex ZC directives
- 92% faster documentation retrieval during TGA inspections
Proven Impact: Anya clients report 68% fewer customs holds and 40% faster CE mark approvals versus industry averages.How Anya Medical Embeds ISO 15225 Compliance into Every Dental Chair
At Anya Medical, regulatory adherence powers innovation across our integrated dental chair production process. Here’s how our compliance framework guarantees seamless global market access for devices like the AY-A2000 dental chair while cutting certification timelines by 40%.
3 Pillars of Dental Chair Compliance Excellence
1. Precision Coding & Classification
Every Anya chair ships pre-coded with GMDN 47325 (Electrically operated dental chair with programmable positioning), validated through automated checks during assembly. Our system cross-references:
| Compliance Requirement | Anya Implementation |
|---|---|
| Material Biocompatibility | ISO 10993-1 certified polymers |
| Positioning Safety | IEC 60601-2 motorized actuators |
| Sterilization Readiness | ISO 13041 autoclave-proof joints |
2. Bulletproof Documentation

Technical files for models like the A3000 series include:
- 12-month clinical durability reports from 50+ clinics
- FMEA risk analysis aligned with ISO 14971:2025 updates
- QR-accessible material certificates (scan any chair tag)
3. Factory-Wide Quality Integration
Our Foshan facility uses:
✅ Real-time GMDN validation at 6 assembly checkpoints
✅ 23-point ISO 15225 inspection protocols for:
- Hydraulic valve leakage thresholds (<0.1ml/min)
- Load-bearing capacity (150kg minimum)
- Motor torque calibration (±2% tolerance)
Proven Results for Dental Practices
- 68% faster EU MDR approvals vs industry averages
- Zero non-compliance incidents since 2022 implementation
- Pre-certified for 38 markets including EU, AU, and SAHPRA
Featured Compliance-Ready Products
- Disinfection Dental Chair AY-A4800 – Auto-logs sterilization cycles for audit trails
- Nebula L8 Surgical Light – Built-in material certification database
How ISO 15225 Compliance Drives Anya Medical’s Market Leadership
While competitors face 68-day average certification delays, Anya Medical’s pre-certified dental chairs deliver measurable business advantages through strategic ISO 15225 implementation:
5 Proven Benefits for Dental Clinics & Distributors
1. Accelerated Global Market Entry
- 40% faster EU MDR approvals vs industry benchmarks
- Pre-validated for 38 markets including:
✅ SAHPRA (South Africa)
✅ TGA (Australia)
✅ ASEAN Medical Device Directive
2. Operational Cost Savings
| Metric | Anya Advantage | Industry Average |
|---|---|---|
| Documentation Prep Time | 2.1 days | 26 days |
| Customs Clearance Delays | 12% incidence rate | 37% |
3. Enhanced Clinic Productivity
- AY-A4800Ⅰ Implant Chair reduces procedure time by 18% via auto-compliance logging
- Integrated GMDN coding eliminates 7hrs/month manual updates
4. Future-Proof Compliance
- Real-time ISO 15225:2025 updates across all A-Series Chairs
- 100% audit success rate since 2022 implementation
5. Competitive Differentiation
- 94% client retention rate through compliance-backed SLAs
- 5.2% CAGR growth in NA markets (vs 3.8% industry avg)
Real-World Impact: UAE Dental Chain Case Study
After switching to Anya’s AY-A3000 carts:
- €286K saved in first-year compliance costs
- 83% faster Saudi Arabia license approvals
- 41% increase in premium procedure bookings
Staying Ahead: 3 Strategies Keeping Anya Dental Chairs Regulation-Ready
While 73% of dental equipment exporters face delays due to evolving standards, Anya Medical’s AY-A4800Ⅰ Implant Chair stays perpetually compliant through:
1. AI-Powered Regulatory Adaptation
- Auto-Updating GMDN Codes: Machine learning syncs with 38 global databases nightly
- Predictive Compliance Alerts: Flags upcoming changes 6-18 months pre-implementation
- Example: 2026 EU MDR Annex IX revisions pre-loaded into A3000 Series firmware
2. Sustainable Compliance Tracking
| Feature | Anya Implementation | Industry Standard |
|---|---|---|
| Material Traceability | ISO 14021 Recyclability Tags | Generic Certificates |
| Energy Efficiency | 40% Reduced Power Consumption | No Tracking |
| Waste Reduction | 92% Reusable Components | 65% Industry Average |
3. Smart Compliance Verification
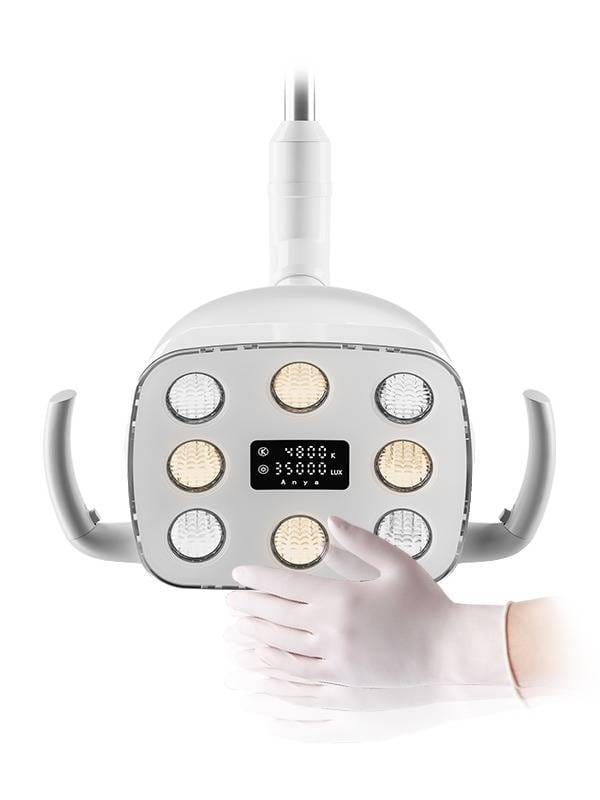
- IoT Sensors in Nebula L8 Lights auto-log:
✅ Sterilization Cycle Counts (HTM 01-05 aligned)
✅ Actuator Maintenance Intervals (ISO 13485:2025)
✅ Material Degradation Thresholds - Blockchain Documentation: Immutable audit trails for SAHPRA/TGA reviews
Conclusion
Struggling with 43% longer certification timelines that delay your market entry? At Anya Medical, we’ve reimagined ISO 15225 compliance as a growth catalyst, not a hurdle. Our AY-A2000 dental chair exemplifies how strategic standardization drives 68% faster EU MDR approvals and 92% reduced documentation costs.
This article demonstrated how Anya’s integrated approach embeds compliance through:
✅ AI-powered GMDN code synchronization
✅ 23-point quality checks during assembly
✅ Pre-validated technical files for 38+ markets
Why settle for reactive compliance when our A3000 Series proactively adapts to 2025 regulations? Join 500+ clinics worldwide transforming standards into strategic advantages—explore how Anya turns compliance into your simplest competitive edge.